Page updated:
October 16, 2021
Author: Emmanuel Boss
View PDF
Introduction to Optical Constituents of the Ocean
Emmanuel Boss, Collin Roesler, and Curtis Mobley contributed to this page.
Classifying the Optically Important Marine Particles
The in-water constituents that affect aquatic optical properties are traditionally lumped into the following categories:
- Sea water (water + inorganic dissolved materials)
- Colored (or chromophoric) dissolved organic material (CDOM)
- Phytoplankton
- Non-phytoplankton organic particles
- Inorganic particles
- Bubbles
The constituents are identified operationally based upon how we measure their optical properties and are often are grouped by like optical properties. For example, the distinction between particulate and dissolved is operationally defined by the filter type or pore size, often a filter pore size of . It is essential to remember that (1) the strict chemical definition is quite different, (2) filter pore size varies from author to author, and (3) it is important to keep track of pore sizes to ensure closure (i.e., don’t define dissolved organic matter by the filtrate of a pore sized filter and then measure particulates on a pore size GF/F filter). Similarly, often all of the non-phytoplankton particles are lumped into a single compartment because their optical properties can be quite similar. Sometimes all of the particulate material is lumped together into suspended particulate material (SPM), or part of it into particulate organic material (POM). This is often done when studying a specific bio-geochemical property using optics.
The differentiation between dissolved and particulate materials (using a filter) does not imply that the dissolved material is organic, though this is most often the assumption. For example, inorganic dissolved substances such as iron oxides (rust) could contribute in certain cases.
A more detailed classification of optically important particles is as follows:
- Biogeochemical classification
- Particulate Organic Material (POC):
- Includes all living or once-living particulate matter such as phytoplankton and detritus.
- Particulate Inorganic Material (PIC):
- Includes all lithogenic and mineralic particulate matter such as sand and calcium carbonate shells, also called Tripton.
- Particulate Material (PM):
- The total organic and inorganic, animate and inanimate particles, also called Seston.
- Biological classification
- Algae:
- Aquatic photosynthetic organisms that can range in size from microscopic (phytoplankton) to tens of meters (giant kelp)
- Phytoplankton:
- Microscopic, single-celled (some form colonies or chains), free-floating, aerobic (live in an oxygenated environment), oxygenic (produce oxygen during photosynthesis) organisms that possess chlorophyll-a and photosynthesize. Some maybe mixotrophic (photosynthetic and heterotrophic). Includes prokaryotes (cyanobacteria) and eukaryotes (diatoms, dinoflagellates, coccolithophorids, and many others).
- Bacterioplankton:
- Microscopic, single-celled (some may form colonies or chains) prokaryotic (no membrane-bound organells) organisms, can be phototrophic (cyanobacteria) or heterotrophic.
- Zooplankton:
- Single- or multi-cellular free-floating heterotrophic organisms that may consume photosynthetic organism or other heterotrophic organisms. They range in size from microscopic to macroscopic (visible by eye). While some are locomotive, they are advected in ocean currents and therefore planktonic.
- Detritus
- Non-living particulate organic matter, including dead bacterial, phytoplankton and zooplankton cells, fragments of cells left from zooplankton grazing, fecal pellets, shells, and marine snow aggregates.
- Size classification
Biological:
- Picoplankton:
- The fraction of plankton between and . Typically phytoplankton.
- Nanoplankton:
- The fraction of plankton between and . Typically phytoplankton and single-celled zooplankton.
- Microplankton:
- The fraction of plankton between and . Typically phytoplankton and single-celled zooplankton.
- Mesoplankton:
- The fraction of plankton between 0.2 mm and 20 mm. Typically multicellular zooplankton, may include large single celled zooplankton and phytoplankton.
Geological:
- Clay:
- The fraction of typically lithogenic or mineralic particles that are less than (0.002 mm).
- Silt:
- The fraction of typically lithogenic or mineralic particles between and (0.002 mm and 0.063 mm). Includes subdivisions fine, medium and coarse.
- Sand:
- The fraction of typically lithogenic or mineralic particles between 0.063 mm () and 2 mm. Includes subdivisions fine, medium and coarse.
- Operational Classification
- Particulate:
- All matter captured by filtration onto a filter of defined nominal pore size. The two most common pore sizes used to identify particles are (e.g., nucleopore filters) or (e.g., glass fiber filters).
- Dissolved:
- All matter that passes through the filter of identified nominal pore size, i.e., filtrate. May include some particulate matter such as very small picoplankton, viruses, and colloids. Optically classified as colored dissolved organic matter (CDOM). Chemists classify colloids as particles in the 1 nm to size range. Marine viruses are typically in the 20-300 nm range.
- Phytoplankton:
- All particulate matter pigmented by phytoplankton-derived pigments, including detrital pigments. Defined by extractive methods using solvents such as acetone and methanol for most pigments and phosphate buffers for phycobilipigments.
- Non-algal particles (NAP):
- The fraction of particulate matter that is not extractable (not a phytoplankton pigment). Includes all living and detrital organic matter such as the non-pigmented portion of phytoplankton cells, detritus, heterotrophic bacteria, and viruses. May also include inorganic mineral particles of both biogenic (e.g., calcite liths and shells) and terrestrial origin (e.g., clay, silt, and sand). Unfortunately, given the current state of the global ocean, we must add tiny particles of plastic to the list of NAPs.
- Particulate Inorganic Matter:
- The fraction of particulate matter that is not organic and therefore remains on the filter after combustion, also called dry ash.
Figure 1 shows the size ranges commonly associated with the different components of natural waters.
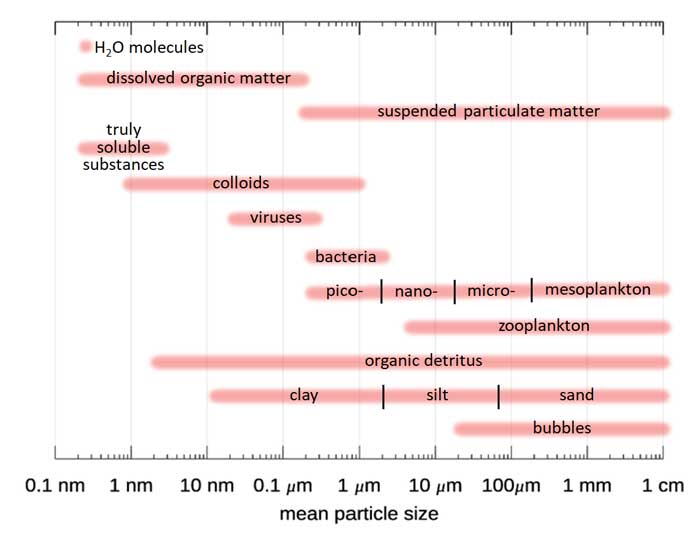
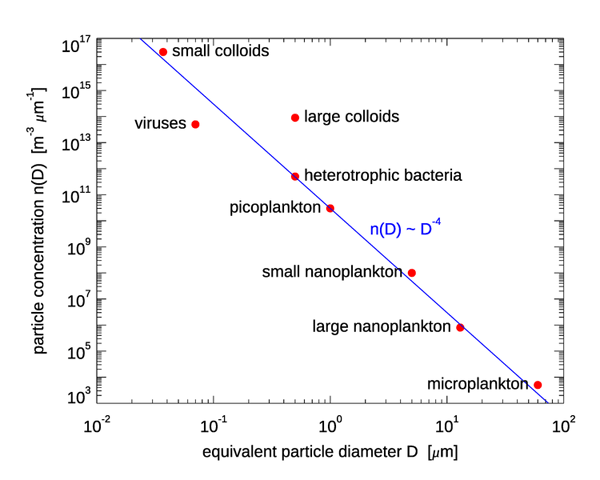
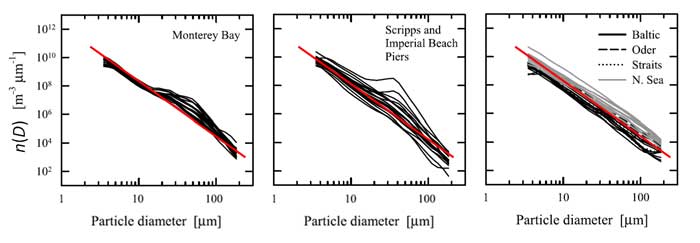
Figure 1 shows the sizes of various particle types. Figure 2 shows how many of different types of biological particles typically occur in marine waters, expressed as a particle size distribution (PSD) . is the diameter of an equivalent-volume sphere, and gives the number of particles per cubic meter, per unit of size range. It must be remembered that such a size distribution may be a good average, but may be quite incorrect for a given time and location. For example, if a phytoplankton bloom is underway, there there will be many more particles of a certain size range, which will put a “bump” in the PSD. In open ocean water, there may be almost no mineral particles. In coastal waters, river inputs or sediment resuspension may make minerals the optically dominant particles. The same can be true in open-ocean water if a coccolithophores are shedding their calcite liths. The blue line shows a power law size distribution with an exponent of -4, i.e., with . A power law distribution with an exponent of -4 is also known as a Junge distribution. Figure 3 shows three sets of PSDs measured in coastal waters. The red lines are Junge size distributions. It is seen that the Junge distribution gives a reasonably good fit over the measured size range, but there are significant deviations from a power law distribution for some of the measurements, and in many cases an power law with a different exponent than -4 gives a better fit.
Modelling the Optical Properties of Marine Particles
Marine particles are extremely complex and varied in their composition, pigmentation, shape, internal structure, and packaging. In order to understand their interaction with light some idealization have to be made. Since it is impractical to study each individual particle (there are more than a billion bacteria in one ml of seawater), particles are lumped into groups of particles having similar properties.
In order to model the optical properties of particles (that is to derive analytical or numerical descriptions of these properties) we need empirical data providing the necessary inputs (size, shape, index of refraction, internal structure and packaging). Some of these data can be obtained from microscopy while others need to be deduced from other measurements; for example to obtain information on the index of refraction of a sediment grain one could immerse it in oils of different indexes and microscopically observe when the least optical contrast (scattering) is observed. Most often an inverse optical approach is observed; that is an optical model (such as Mie theory) is used to fit empirical data and the values of the index of refraction that provide the best match with the observations are chosen to be those of the suspension. Aas (1996), Table 1, provides a compilation for data on the real part of index of refraction of marine particles and the different methodologies used to obtain them. He also shows how the estimates for phytoplankton are consistent with a model of the index of refraction that is based on their composition.
The specific models for the different types particles are provided on their own pages. They vary in methodology used to obtain them; some are based strictly on observations, either in the lab or in the field. Other are based on numerical calculations (e.g. using Mie theory) with inputs obtained from observations. The latter are particularly useful when laboratory observations are lacking due to the complexity associated with the measurements (e.g. the volume scattering function). It is particularly satisfying when independent observations and models agree (referred to as optical closure), which provides mutual validation for the approaches.
The next few pages examine the optical properties of various constituents. Commonly used bio-optical models are surveyed in Level 2.

 See comments posted for this page and leave your own.
See comments posted for this page and leave your own.